Home>Science>The Mind-Blowing Secret Behind Covalent Bonds Revealed!

Science
The Mind-Blowing Secret Behind Covalent Bonds Revealed!
Published: January 19, 2024
Uncover the mind-blowing secret behind covalent bonds in this fascinating exploration of science. Gain a deeper understanding of the intricate world of chemical bonding.
(Many of the links in this article redirect to a specific reviewed product. Your purchase of these products through affiliate links helps to generate commission for Noodls.com, at no extra cost. Learn more)
Table of Contents
Introduction
Welcome to the fascinating world of covalent bonds! Have you ever wondered how atoms form strong connections with one another to create the countless substances that make up our world? The answer lies in the captivating realm of chemistry, where the concept of covalent bonding takes center stage.
In this article, we will embark on an enlightening journey to unravel the profound mysteries behind covalent bonds. By delving into the intricate interactions between atoms, we will uncover the awe-inspiring secret that governs the formation of these bonds. Prepare to be amazed as we unveil the hidden forces that bind atoms together and shape the very fabric of our physical reality.
Join me as we explore the profound implications and practical applications of covalent bonds, shedding light on their pivotal role in diverse fields, from pharmaceuticals and materials science to environmental studies and beyond. Together, we will gain a deeper understanding of this fundamental concept and its far-reaching impact on the world around us.
So, fasten your seatbelts and get ready to embark on an exhilarating journey into the heart of covalent bonding. By the end of this exploration, you will emerge with a newfound appreciation for the remarkable forces that underpin the structure and function of matter as we know it. Let's embark on this captivating odyssey to unlock the mind-blowing secret behind covalent bonds!
Understanding Covalent Bonds
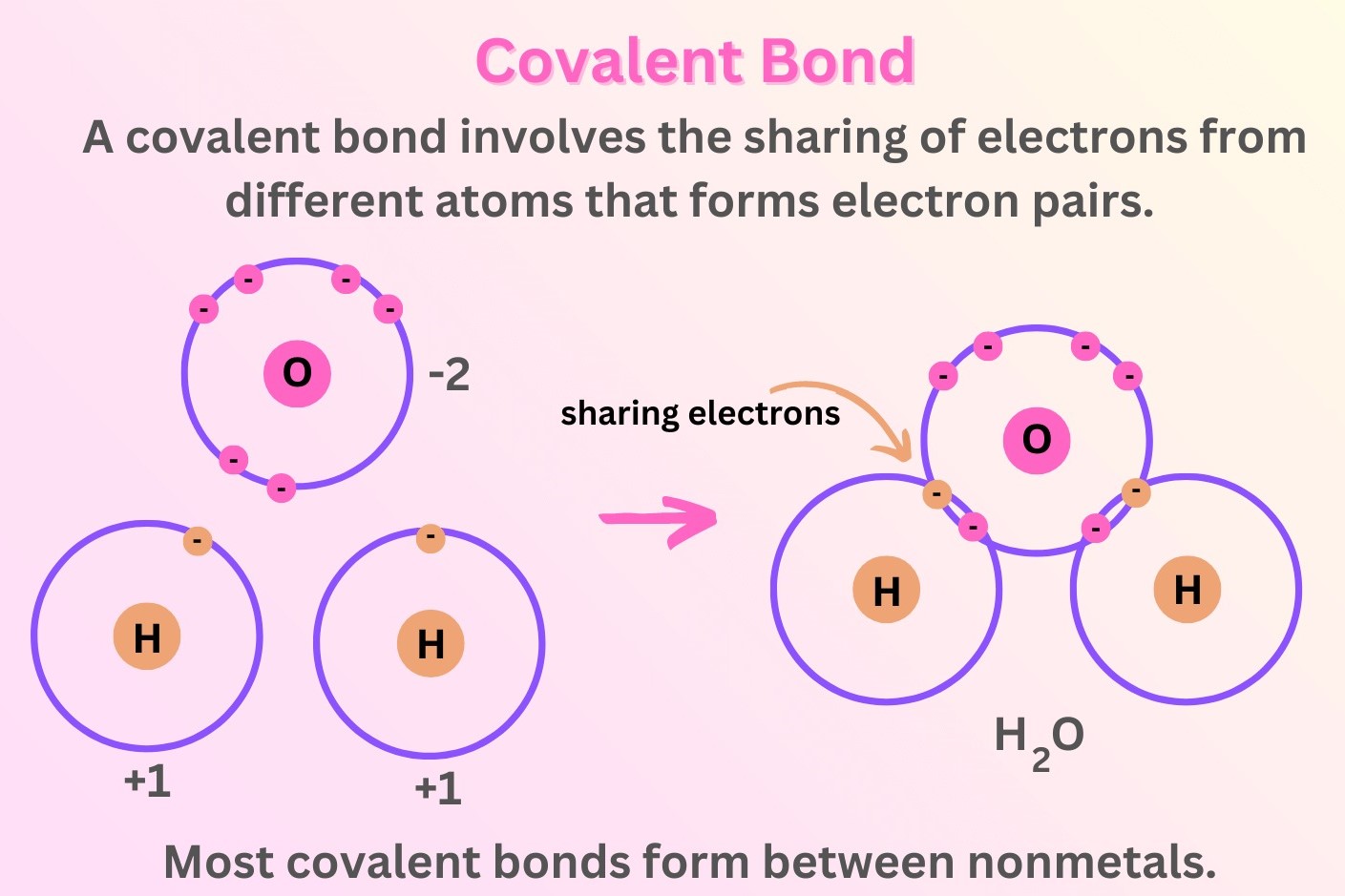
Covalent bonds, one of the fundamental concepts in chemistry, play a pivotal role in shaping the properties of various substances. At the heart of a covalent bond lies the sharing of electron pairs between atoms, leading to the formation of a strong connection. This sharing of electrons enables atoms to achieve a stable configuration, typically resembling the noble gas configuration, characterized by a full outer electron shell.
In a covalent bond, atoms share electrons in a manner that allows each atom to attain a more stable electron configuration. This sharing of electrons results in the formation of a molecular structure, where atoms are held together by the mutual attraction of the shared electrons. Unlike ionic bonds, where electrons are transferred from one atom to another, covalent bonds involve a cooperative sharing of electrons, creating a balanced distribution of charge within the molecule.
The strength of a covalent bond is determined by the sharing of electrons and the distance between the nuclei of the bonded atoms. The sharing of electrons is influenced by factors such as the number of shared electron pairs and the electronegativity of the atoms involved. Additionally, the spatial arrangement of atoms within the molecule, known as the molecular geometry, contributes to the overall stability and reactivity of the compound.
Covalent bonds can be further classified into polar and nonpolar bonds based on the electronegativity difference between the bonded atoms. In polar covalent bonds, the electrons are unequally shared, leading to a partial separation of charge within the molecule. This results in the development of a dipole moment, where one end of the molecule possesses a slight negative charge while the other end exhibits a partial positive charge. On the other hand, nonpolar covalent bonds involve an equal sharing of electrons, resulting in a symmetrical distribution of charge within the molecule.
Understanding the nature of covalent bonds is essential for comprehending the behavior of various compounds, including organic molecules, polymers, and biological macromolecules. The unique properties exhibited by covalently bonded substances, such as their solubility, reactivity, and structural stability, are intricately linked to the characteristics of the covalent bonds within the molecules.
As we continue our exploration of covalent bonds, we will delve deeper into the mind-blowing secret that governs the formation of these remarkable connections, shedding light on the captivating forces that shape the world of chemistry. Join me as we unravel the mysteries of covalent bonding and uncover its profound implications across diverse scientific disciplines.
The Mind-Blowing Secret
At the heart of the enigmatic realm of covalent bonds lies a mind-blowing secret that governs the very essence of chemical interactions. This secret is none other than the remarkable concept of electron orbitals and molecular orbitals, which underpin the formation and stability of covalent bonds.
Electron orbitals, often depicted as three-dimensional regions where electrons are likely to be found, are crucial in understanding the behavior of electrons within an atom. These orbitals, characterized by distinct shapes and orientations, provide a framework for visualizing the distribution of electrons around the atomic nucleus. The unique arrangement of electron orbitals within an atom influences its reactivity and bonding capabilities, setting the stage for the formation of covalent bonds.
When atoms approach each other to form a covalent bond, their electron orbitals undergo a mesmerizing transformation, giving rise to molecular orbitals. These molecular orbitals represent the combined distribution of electrons from the participating atoms, creating a new entity that transcends the individual atoms themselves. The merging of electron orbitals leads to the formation of bonding and antibonding molecular orbitals, each with its distinct energy levels and spatial characteristics.
The mind-blowing secret behind covalent bonds lies in the delicate interplay between these bonding and antibonding molecular orbitals. As atoms unite to share electrons and establish a stable configuration, the bonding molecular orbitals facilitate the formation of a strong, cohesive bond, anchoring the atoms together within the molecule. Conversely, the antibonding molecular orbitals introduce a fascinating dimension of electron interactions, contributing to the overall stability and energy profile of the covalently bonded system.
The mind-blowing secret unravels the intricate dance of electrons as they navigate through the molecular orbitals, shaping the unique properties and reactivity of covalently bonded compounds. This captivating revelation underscores the profound influence of electron behavior on the stability and structure of molecules, illuminating the underlying forces that govern the world of chemistry.
By delving into the mesmerizing realm of electron orbitals and molecular orbitals, we gain a deeper appreciation for the mesmerizing intricacies that define covalent bonds. The mind-blowing secret reveals the awe-inspiring orchestration of electron interactions that underpin the formation of these fundamental connections, offering a glimpse into the captivating world of chemical bonding.
As we uncover the mind-blowing secret behind covalent bonds, we gain a newfound perspective on the intricate forces that shape the molecular landscape, paving the way for groundbreaking discoveries and innovations across diverse scientific domains. Join me as we unravel the captivating mysteries of electron orbitals and molecular orbitals, unlocking the hidden secrets that define the essence of covalent bonding.
Implications and Applications
The profound implications of covalent bonds extend far beyond the realm of theoretical chemistry, permeating diverse scientific disciplines and practical applications. Understanding the intricate nature of covalent bonds unlocks a wealth of opportunities for innovation and discovery, shaping the landscape of materials science, pharmaceutical development, and environmental studies.
In materials science, the unique properties of covalently bonded substances have revolutionized the design and engineering of advanced materials with tailored functionalities. From high-performance polymers and semiconductors to novel nanomaterials, the stability and structural diversity conferred by covalent bonds have paved the way for groundbreaking advancements in electronics, aerospace technologies, and sustainable energy solutions. The precise control over molecular structures afforded by covalent bonding has fueled the development of innovative materials with enhanced mechanical strength, thermal stability, and electrical conductivity, opening new frontiers in material design and manufacturing.
In the pharmaceutical industry, the understanding of covalent bonds has catalyzed the discovery and development of life-saving drugs and therapeutic agents. By harnessing the principles of covalent bonding, researchers have unlocked novel strategies for designing targeted pharmaceutical compounds that exhibit enhanced efficacy and specificity. The precise modulation of covalent interactions within drug molecules has led to the creation of potent pharmaceuticals with improved bioavailability and reduced off-target effects, offering new hope for addressing complex diseases and unmet medical needs.
In environmental studies, the implications of covalent bonds resonate in the realm of pollutant remediation and sustainable technologies. The application of covalent bonding principles in environmental engineering has facilitated the design of innovative strategies for pollutant sequestration, water purification, and air quality management. By leveraging the unique reactivity and stability of covalently bonded compounds, scientists have devised eco-friendly solutions for mitigating environmental contaminants and developing sustainable materials with minimal ecological impact, ushering in a new era of environmental stewardship and resource conservation.
The far-reaching implications and practical applications of covalent bonds underscore their pivotal role in driving scientific progress and technological innovation. From the development of advanced materials and pharmaceutical breakthroughs to environmental sustainability initiatives, the profound understanding of covalent bonding continues to shape the forefront of scientific research and industrial advancements, offering boundless opportunities for addressing complex challenges and improving the quality of life for humanity.
As we contemplate the implications and applications of covalent bonds, we are reminded of the enduring impact of these fundamental connections on the fabric of our modern world. The remarkable versatility and transformative potential of covalent bonding serve as a testament to the enduring legacy of scientific inquiry and the boundless possibilities that emerge from unraveling the mysteries of the natural world.
Conclusion
In conclusion, the captivating journey into the realm of covalent bonds has unveiled the profound secrets that govern the formation and behavior of these fundamental connections. From the intricate dance of electron orbitals to the transformative implications across scientific disciplines, the essence of covalent bonding transcends the boundaries of theoretical chemistry, shaping the forefront of materials science, pharmaceutical innovation, and environmental stewardship.
As we reflect on the mind-blowing secret behind covalent bonds, we are reminded of the remarkable synergy between scientific exploration and real-world applications. The understanding of covalent bonding has empowered researchers and innovators to engineer advanced materials with unprecedented properties, revolutionize drug discovery and development, and pioneer sustainable technologies that safeguard our environment.
The implications of covalent bonds extend beyond the confines of laboratory experiments, resonating in the products we use, the medicines that save lives, and the solutions that address environmental challenges. The enduring legacy of covalent bonding lies in its ability to inspire curiosity, drive discovery, and foster a deeper appreciation for the intricate forces that shape our physical world.
As we embark on future scientific endeavors, the revelations of covalent bonding serve as a testament to the boundless potential of human ingenuity and the enduring quest to unravel the mysteries of nature. With each new discovery, we inch closer to harnessing the full potential of covalent bonds, unlocking innovative solutions to complex problems and forging a path towards a brighter, more sustainable future for generations to come.
In the grand tapestry of scientific exploration, the mind-blowing secret behind covalent bonds stands as a testament to the awe-inspiring beauty of the natural world and the limitless possibilities that emerge from understanding its inner workings. Let us continue to embrace the spirit of inquiry, embarking on new adventures that unravel the mysteries of covalent bonding and propel humanity towards greater heights of knowledge and innovation.