
Science
The Surprising Truth About CH3COO!
Published: January 16, 2024
Uncover the surprising truth about CH3COO in the world of science. Explore the fascinating insights and discoveries in this intriguing field.
(Many of the links in this article redirect to a specific reviewed product. Your purchase of these products through affiliate links helps to generate commission for Noodls.com, at no extra cost. Learn more)
Table of Contents
Introduction
CH3COO, also known as acetate, is a fascinating and versatile chemical compound that plays a crucial role in various aspects of our lives. Despite its seemingly unassuming nature, CH3COO possesses remarkable properties and applications that are often overlooked. In this article, we will delve into the intriguing world of CH3COO, uncovering its true significance and dispelling common misconceptions surrounding this compound.
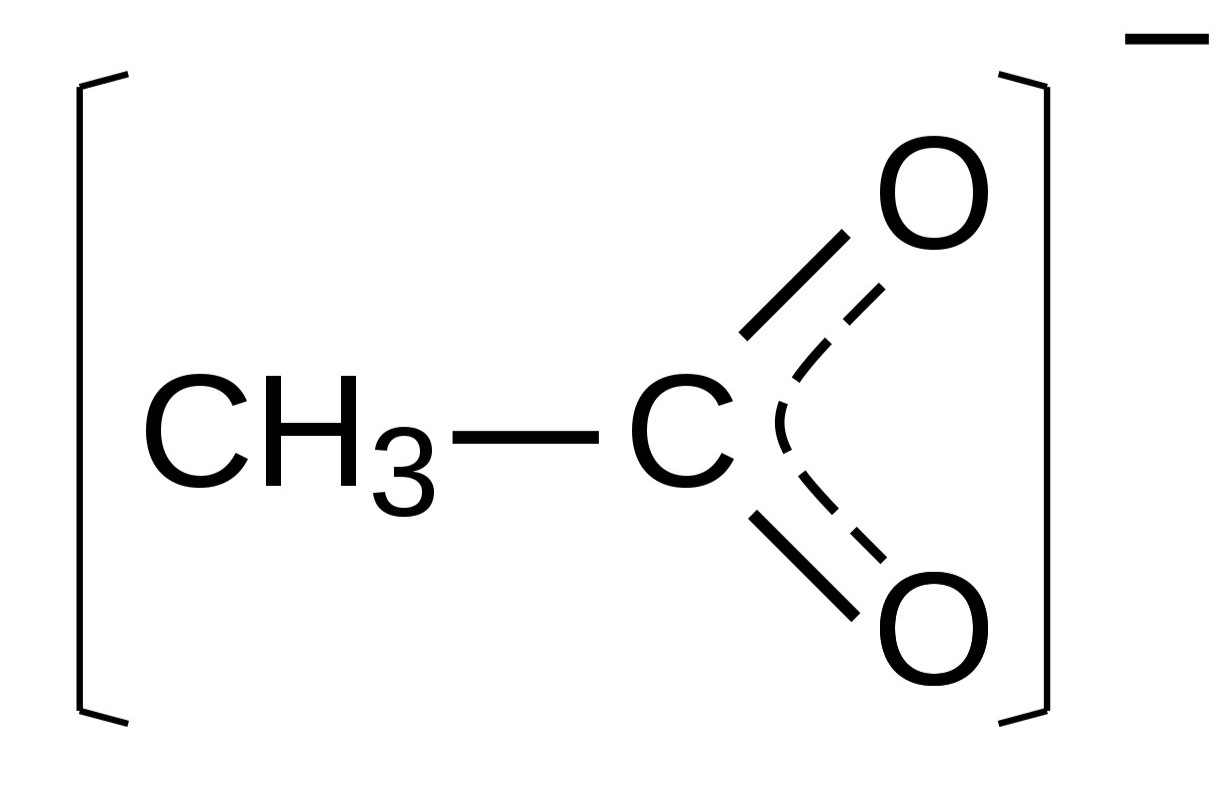
Acetate, with its molecular formula CH3COO-, is the conjugate base of acetic acid. This simple yet impactful compound is widely utilized in diverse fields, ranging from chemistry and biochemistry to industry and everyday products. By exploring the properties and applications of CH3COO, we can gain a deeper understanding of its importance and the pivotal role it plays in shaping our modern world.
Stay tuned as we unravel the surprising truth about CH3COO, shedding light on its multifaceted nature and unveiling the hidden potential that lies within this unassuming compound. Get ready to embark on a journey of discovery as we uncover the remarkable properties and applications of CH3COO, challenging preconceived notions and unveiling the true essence of this extraordinary chemical compound.
What is CH3COO?
CH3COO, also known as acetate, is a crucial chemical compound that holds immense significance in various scientific, industrial, and everyday contexts. It is the acetate ion, represented by the chemical formula CH3COO-, and is derived from acetic acid. This simple yet versatile compound plays a pivotal role in numerous chemical reactions and processes, making it an indispensable component in a wide array of applications.
Acetate is a key player in the realm of organic chemistry, where it serves as a fundamental building block for the synthesis of numerous organic compounds. Its presence is ubiquitous in nature, as it is a common constituent of many natural products, including fruits, wine, and vinegar. In addition to its natural occurrence, acetate is also extensively utilized in laboratory settings, where it serves as a valuable reagent in various experimental procedures.
Furthermore, the acetate ion plays a crucial role in biochemical processes within living organisms. It is involved in the metabolism of carbohydrates and fats, contributing to the production of energy essential for cellular functions. Acetate also participates in the biosynthesis of essential biomolecules, highlighting its significance in sustaining life processes.
Beyond its role in chemistry and biochemistry, acetate finds widespread application in industrial processes and consumer products. It serves as a precursor for the production of various chemicals, including solvents, polymers, and pharmaceuticals. Moreover, acetate compounds are utilized in the manufacturing of textiles, plastics, and food additives, underscoring their diverse industrial applications.
In the realm of environmental science, acetate plays a notable role in the natural carbon cycle. It is involved in the degradation of organic matter in aquatic and terrestrial environments, contributing to the recycling of carbon compounds in nature. This pivotal role in the carbon cycle highlights the ecological significance of acetate in maintaining environmental balance.
In summary, CH3COO, or acetate, is a multifaceted compound with far-reaching implications in diverse fields. Its pivotal role in organic chemistry, biochemistry, industry, and the environment underscores its significance as a fundamental building block and a versatile component in various applications. By understanding the true nature of acetate and its wide-ranging impact, we can gain a deeper appreciation for the remarkable versatility and importance of this unassuming chemical compound.
Common Misconceptions
Common misconceptions surrounding CH3COO, or acetate, often stem from its perceived simplicity and ubiquitous presence in everyday products. One prevalent misconception is the underestimation of the significance of acetate in chemical reactions and industrial processes. Due to its widespread use in consumer products and its natural occurrence in various foods and beverages, acetate is often overlooked as a mere byproduct or additive, leading to the misconception that it lacks substantial chemical or industrial importance.
Another misconception pertains to the environmental impact of acetate compounds. While acetate is naturally present in the environment and is involved in the carbon cycle, there is a misconception that its widespread use in consumer products and industrial processes has negligible environmental consequences. This misconception overlooks the potential implications of acetate release into the environment, particularly in the context of waste disposal and industrial emissions.
Furthermore, there is a common misconception regarding the safety and toxicity of acetate compounds. Some individuals may perceive acetate as inherently harmless due to its natural occurrence in food and beverages, overlooking the potential hazards associated with certain acetate derivatives and industrial applications. This misconception underscores the need for a comprehensive understanding of the safety considerations and potential risks associated with acetate compounds in various contexts.
Additionally, there is a misconception surrounding the perceived limitations of acetate in terms of its industrial applications. Some may underestimate the versatility of acetate compounds and their significance as precursors for the production of various chemicals, polymers, and materials. This misconception fails to recognize the wide-ranging impact of acetate in industrial processes and its role in the synthesis of essential products that permeate numerous sectors.
Addressing these common misconceptions is essential in fostering a more accurate understanding of the significance and implications of acetate in various domains. By dispelling these misconceptions, we can unveil the true complexity and importance of acetate, paving the way for a more comprehensive appreciation of its multifaceted nature and pervasive influence in science, industry, and everyday life.
The Properties of CH3COO
CH3COO, or acetate, exhibits a diverse range of properties that contribute to its widespread significance in various scientific, industrial, and environmental contexts. Understanding these properties is essential for comprehending the multifaceted nature of CH3COO and its pivotal role in numerous applications.
-
Chemical Structure: The acetate ion, represented by the chemical formula CH3COO-, consists of two carbon atoms, three hydrogen atoms, and two oxygen atoms. Its structure embodies a unique combination of covalent bonds, imparting stability and reactivity to the compound. This molecular arrangement serves as the foundation for the diverse chemical properties of acetate.
-
Acidity and Basicity: Acetate exhibits amphiprotic behavior, functioning as both an acid and a base in chemical reactions. As the conjugate base of acetic acid, acetate can accept a proton, thereby displaying basic characteristics. Conversely, in the presence of stronger bases, acetate can donate a proton, showcasing acidic properties. This dual nature enables acetate to participate in a wide array of chemical processes.
-
Solubility: Acetate compounds are generally soluble in water, contributing to their widespread use in aqueous solutions and chemical reactions. This high solubility facilitates the incorporation of acetate into various industrial processes and biochemical reactions, making it a versatile component in solution chemistry.
-
Stability and Reactivity: The stability of acetate compounds, coupled with their reactivity in specific chemical environments, underscores their utility in organic synthesis and industrial applications. Acetate derivatives can undergo diverse chemical transformations, serving as crucial intermediates in the production of pharmaceuticals, polymers, and specialty chemicals.
-
Environmental Impact: Acetate compounds play a significant role in the natural carbon cycle, contributing to the decomposition of organic matter and the recycling of carbon compounds in the environment. Their presence in natural ecosystems highlights their ecological importance and their influence on environmental processes.
-
Industrial Applications: The properties of acetate, including its solubility, stability, and reactivity, render it indispensable in the manufacturing of various products. From textiles and plastics to pharmaceuticals and food additives, acetate compounds serve as essential components in a wide range of industrial processes.
Understanding the diverse properties of CH3COO is crucial for recognizing its pervasive influence and multifaceted applications. By comprehending the chemical, physical, and environmental properties of acetate, we can gain a deeper appreciation for its significance in shaping the modern world and driving advancements across numerous domains.
Applications of CH3COO
The versatile nature of CH3COO, or acetate, is exemplified through its wide-ranging applications across diverse domains, encompassing chemistry, biochemistry, industry, and environmental science. The inherent properties of acetate, coupled with its reactivity and compatibility with various processes, render it indispensable in numerous applications.
Organic Synthesis
Acetate serves as a fundamental building block in organic synthesis, where it is utilized in the production of a myriad of organic compounds. Its role as a versatile precursor enables the synthesis of pharmaceuticals, agrochemicals, and specialty chemicals, contributing to advancements in medicine, agriculture, and materials science. The incorporation of acetate derivatives in organic reactions underscores their pivotal role in driving innovation and the development of novel compounds with diverse functionalities.
Industrial Processes
In industrial settings, acetate compounds find extensive application in the manufacturing of textiles, plastics, and various consumer products. Acetate-based materials, such as cellulose acetate, are utilized in the production of fibers, films, and coatings, contributing to the textile and packaging industries. Additionally, acetate derivatives serve as essential components in the formulation of adhesives, solvents, and specialty chemicals, further highlighting their significance in industrial processes and product development.
Environmental Remediation
The environmental significance of acetate is evident in its applications in environmental remediation and waste treatment. Acetate compounds are utilized in bioremediation processes to facilitate the degradation of organic pollutants in soil and water systems. Their involvement in promoting the natural breakdown of contaminants underscores their role in mitigating environmental pollution and contributing to sustainable remediation strategies.
Biochemical Pathways
In biochemistry, acetate plays a vital role in metabolic pathways and cellular processes within living organisms. It serves as a substrate for energy production and the biosynthesis of essential biomolecules, contributing to the metabolic activities of cells. Acetate also participates in the acetylation of proteins, influencing their structure and function. The diverse biochemical roles of acetate underscore its significance in sustaining life processes and cellular functions.
Food and Beverage Industry
Acetate compounds are utilized in the food and beverage industry as preservatives, flavoring agents, and acidity regulators. They contribute to the preservation of food products, the enhancement of flavors, and the maintenance of pH balance in various culinary applications. The incorporation of acetate in food and beverage production underscores its role in ensuring product quality and safety.
Pharmaceutical Applications
In the pharmaceutical industry, acetate derivatives are employed in the formulation of medications, serving as excipients, solubilizing agents, and drug delivery vehicles. Their compatibility with pharmaceutical formulations and their inert nature make acetate compounds essential in the development of pharmaceutical products, thereby contributing to the advancement of healthcare and therapeutic treatments.
The diverse applications of CH3COO underscore its pervasive influence and indispensable role across various sectors. From organic synthesis and industrial manufacturing to environmental remediation and biochemical processes, acetate compounds continue to shape advancements in science, technology, and everyday products, exemplifying their enduring significance in modern society.
Conclusion
In conclusion, the multifaceted nature of CH3COO, or acetate, transcends its seemingly simple chemical composition, revealing a compound of immense significance and versatility. Throughout this exploration, we have uncovered the surprising truth about acetate, dispelling common misconceptions and shedding light on its pivotal role in various domains.
From its fundamental properties to its diverse applications, acetate has proven to be an indispensable component in organic synthesis, industrial processes, environmental remediation, biochemical pathways, and the production of consumer goods. Its role as a building block for organic compounds, coupled with its compatibility with diverse chemical processes, underscores its enduring importance in driving innovation and advancements across numerous sectors.
Furthermore, the environmental impact of acetate compounds cannot be overlooked, as they contribute to the natural carbon cycle and play a crucial role in maintaining environmental balance. The involvement of acetate in bioremediation processes highlights its potential in mitigating environmental pollution and fostering sustainable remediation strategies.
In the realm of biochemistry, acetate's role in metabolic pathways and cellular functions underscores its significance in sustaining life processes, further emphasizing its pervasive influence in biological systems.
Moreover, the applications of acetate in the food and beverage industry, pharmaceutical formulations, and industrial manufacturing underscore its enduring relevance in ensuring product quality, advancing healthcare, and driving the production of essential consumer goods.
By unraveling the surprising truth about CH3COO, we have gained a deeper appreciation for the remarkable versatility and importance of this unassuming compound. Its impact spans across scientific, industrial, and everyday contexts, shaping our modern world and contributing to advancements in diverse fields.
As we conclude this exploration, it is evident that acetate, with its multifaceted properties and wide-ranging applications, stands as a testament to the enduring significance of seemingly simple compounds in driving progress and innovation. Embracing the true essence of acetate allows us to recognize its pivotal role in shaping our world and inspiring future developments across various domains.